Join-us
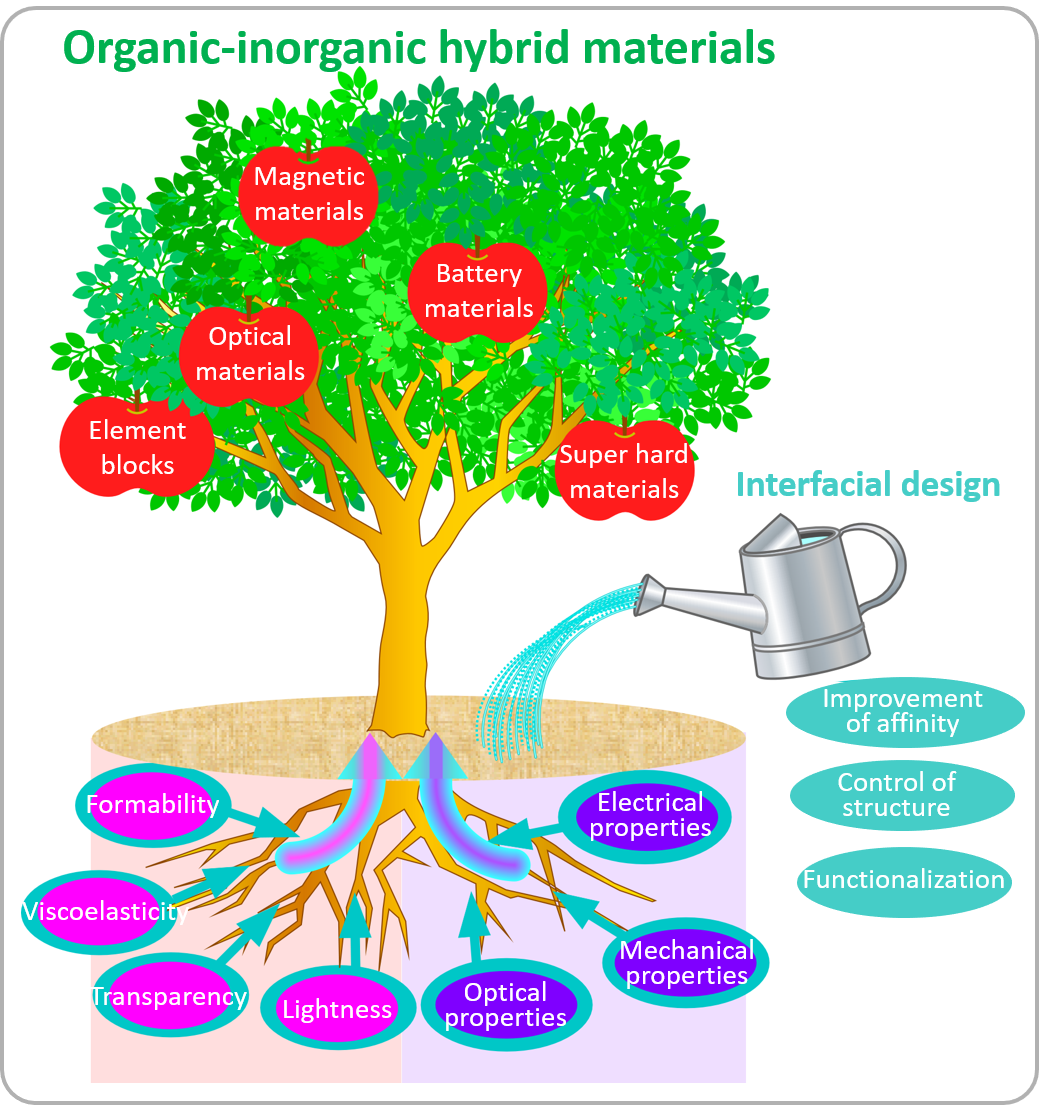
Chemistry provides various materials to support our lives. New materials promote advances in technologies, and advanced technologies can improve our life in many ways. Sugahara laboratory contributes to this improvement through its primary focus on creation of new organic-inorganic hybrid materials.
How to prepare excellent organic-inorganic hybrid materials
The term “hybrid” was originally used to mean something like “crossbred” for plants and animals. In the fields of science and technology, on the other hand, “hybrid” refers to a product which is fabricated using two or more technologies or a machine which is composed of two different types of parts (“hybrid car” is a well-known example). In the field of materials chemistry in particular, “hybrid” is applied to materials comprising two or more components, generally inorganic and organic components. Although both inorganic and organic materials have been widely used in various fields, the properties achievable using inorganic or organic materials alone are limited. Thus, organic-inorganic hybrid materials that can combine the properties of both inorganic and organic materials are expected to become “next generation materials.” Since the possible combinations are infinite, creation of vast numbers of new materials with variable properties is expected. Their properties will not be governed only by the volume ratio of inorganic and organic materials; they will also depend largely on sizes of their inorganic and organic components as well as the interfaces between them. Thus, techniques to prepare organic-inorganic hybrid materials with controlled sizes and component interfaces should be the key to providing them with excellent properties.
Approaches to preparation of organic-inorganic hybrid materials
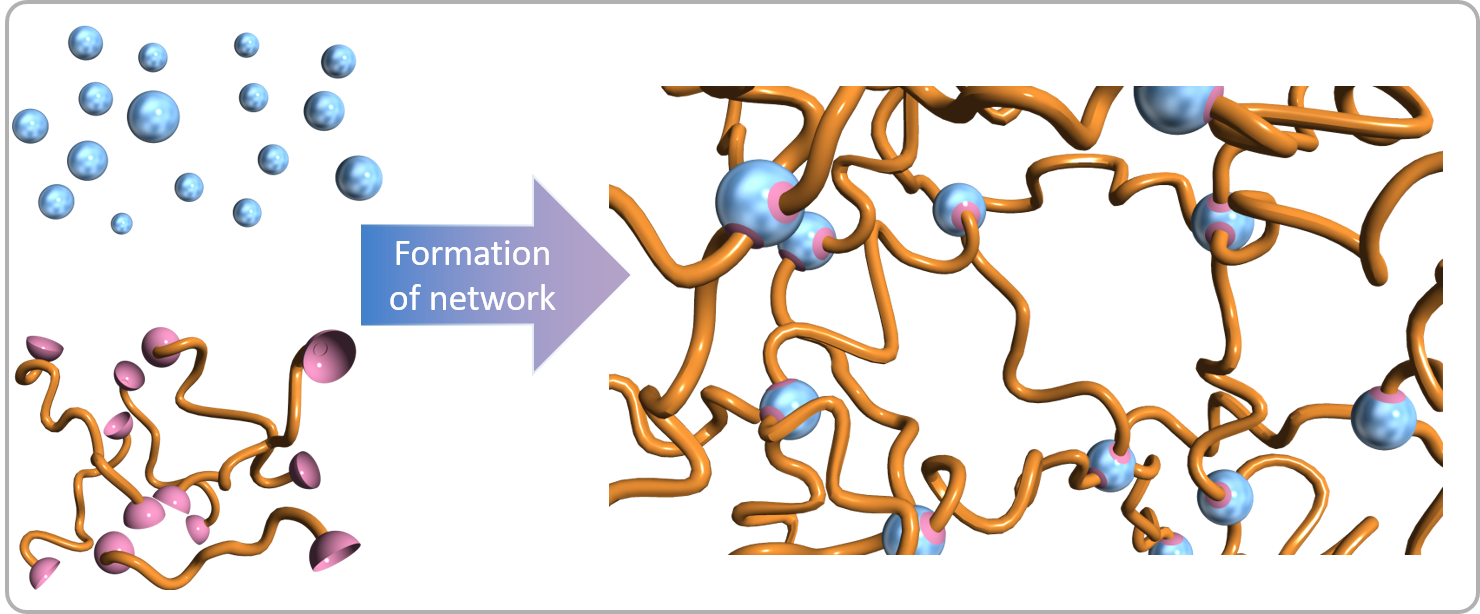
We currently have two approaches to preparation of organic-inorganic hybrid materials. Organic-inorganic hybrid materials using inorganic nanomaterials: To enable the use of inorganic nanomaterials in organic-inorganic hybrid materials, methods of immobilizing organic groups on their surfaces have been developed. Organic molecules and polymers are immobilized on the surfaces of inorganic nanomaterials, which are mainly metal oxide nanoparticles and nanosheets, via covalent bonds. If necessary, organic molecules are prepared by organic synthetic methods, or polymer chains are grown from functional groups immobilized on inorganic surfaces. Metal-oxygen-phosphorus bonds are mainly used at the interfaces between the organic groups and inorganic surfaces. We have developed organic-inorganic hybrid materials by embedding such surface-modified nanoparticles in polymer matrices. We have also develop their uses as nanomaterials Organic-inorganic hybrid materials using bottom-up processes: Organic-inorganic hybrid materials have also been prepared using the reactions of molecules bearing several -P(O)(OH)2 groups, such as organodiphosphonic acid with a metal compound. When organodiphoshponic acids ((OH)2(O)P-R-P(O)(OH)2) bearing R groups with functionality are employed, for example, functional organic-inorganic hybrid materials can be obtained. Networks with M-O-P bonds are formed using metal compounds such as metal chlorides.
Preparation of inorganic materials using organic-inorganic hybrid materials
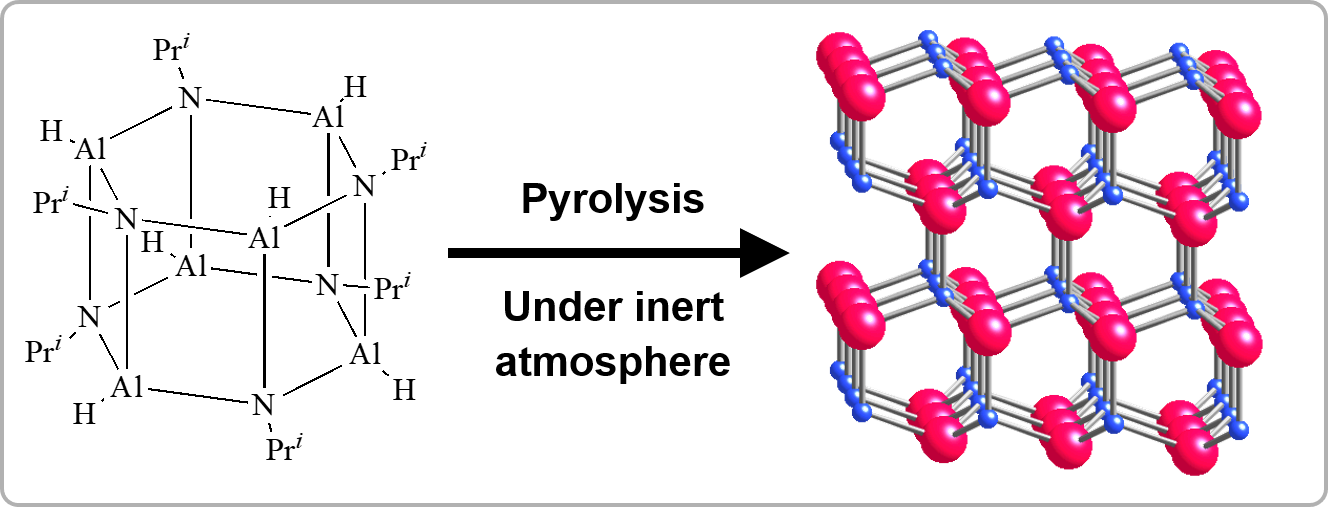
As for uses totally distinct from the above-mentioned use of organic-inorganic hybrid materials, their use as precursors for ceramic materials has been developed. Although ceramic materials are generally prepared by powder processing involving sintering, this method cannot be applied to every shape. Among organic-inorganic hybrid materials, the compounds classified as inorganic polymers, which exhibit basic polymer properties, can be utilized as precursors for designing molecular structures to obtain ceramic materials by eliminating organic components through pyrolysis or hydrolysis. Typically, polymers bearing metal-nitrogen and metal-carbon networks are used as “preceramic polymers,” and the non-oxide ceramics obtained are called “polymer-derived ceramics (PDCs).” This process is also capable of preparing ceramic thin films and fibers, which cannot be obtained via a powder process. In addition, by using amorphous materials that can be prepared by pyrolysis at relatively lower temperatures as intermediate, superhard materials which are the high-pressure phases can be prepared by treatment at high temperatures and high pressures that are less extreme than those required for the conversion of crystals.
Our research strategy
- In order to prepare target functional materials, we combine synthetic techniques if necessary. These techniques are not limited to synthetic techniques in inorganic synthetic chemistry but also include those in organic synthetic chemistry, organometal chemistry and polymer chemistry. Although techniques are categorized in different fields, we consider the use of various synthetic techniques important.
- We believe characterization of prepared materials is important. Thus, we focus on revealing the structures and functionalities of materials by the use of various analytical techniques. When in-campus characterization is insufficient, characterization is achieved through collaboration with external experts.
- We focus mainly on preparation of materials and their characterization. If necessary to reveal the functionalities of prepared materials, we collaborate with other research groups in academia and industry. We discuss the properties of materials based on their structures as revealed by our characterization techniques for the design of materials with better functionalities. We also employ these collaborations with researchers in academic and industrial groups, to learn the requirements of materials for their social implementation.